
Fundamentals of Chemistry
Chemistry is a scientific discipline that studies matter, its properties, and the changes it undergoes during chemical reactions. It explores the fundamental building blocks of the universe, including atoms, molecules, and their interactions.
Chemistry seeks to understand the behaviour of substances, both natural and synthetic, at the molecular and atomic levels. It examines how atoms combine to form molecules and how these molecules interact with one another to create new substances.
The periodic table of elements
The periodic table of elements is a systematic arrangement of all known chemical elements. It organizes elements based on their atomic number, electron configuration, and recurring chemical properties. The table provides a comprehensive framework that allows scientists to understand the relationships, trends, and behavior of elements.
The periodic table consists of rows called periods and columns called groups. Each element is represented by a unique symbol and is placed in a specific location within the table. The elements are organized in ascending order of atomic number, which corresponds to the number of protons in an atom’s nucleus.
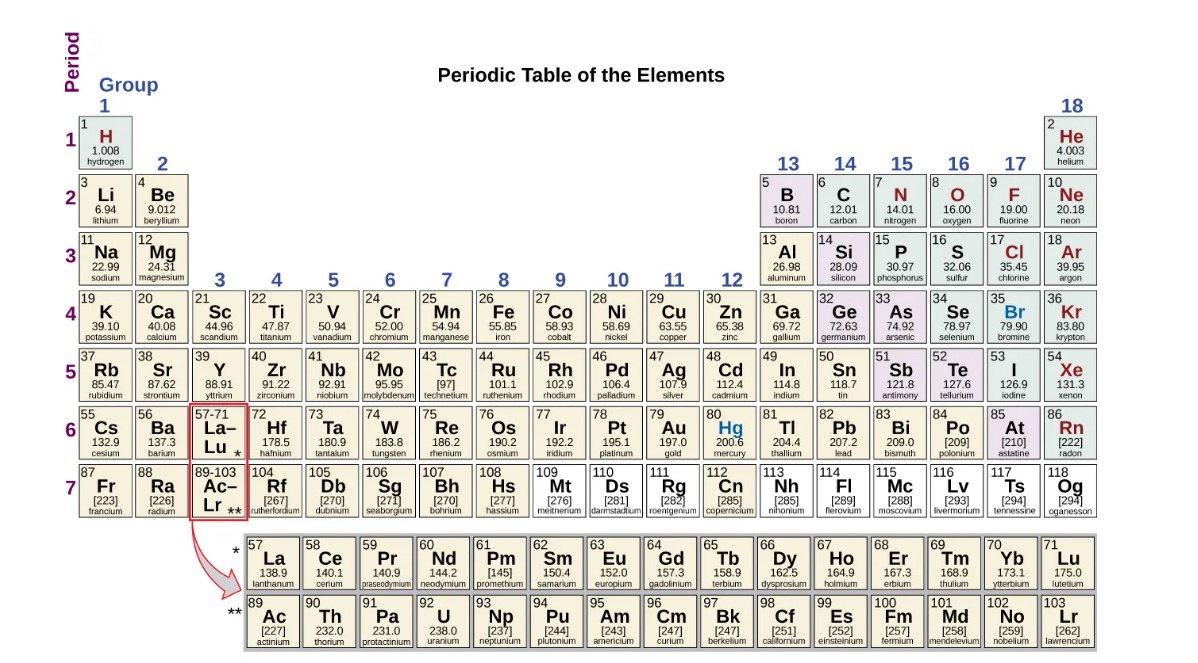


Flowers, P., Neth, E., Robinson, W., Theopold, K., & Langley, R. (2019). The Periodic Table. In Chemistry: Atoms First 2e. OpenStax.
Access for free at https://openstax.org/books/chemistry-atoms-first-2e/pages/1-introduction
Atoms
An atom is the basic building block of matter, representing the smallest unit of a chemical element that retains its unique properties. At the core of every atom is a central nucleus that is composed of positively charged protons and uncharged neutrons. Surrounding the nucleus are negatively charged electrons that occupy regions called electron shells or energy levels.





